- #Are hydrophobic amino acids polar or nonpolar code
- #Are hydrophobic amino acids polar or nonpolar free
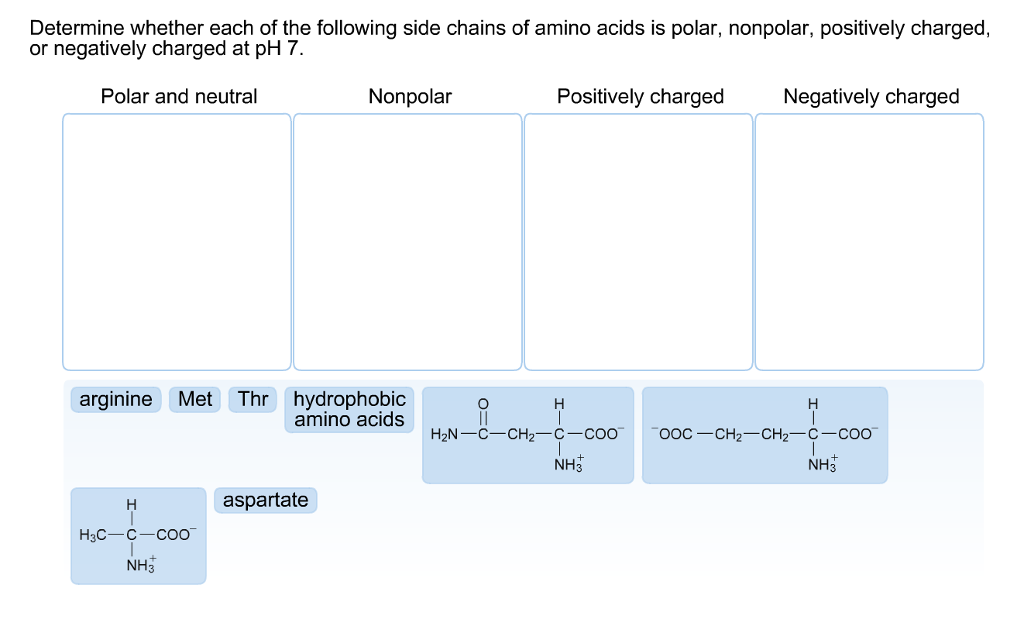
Hydrophobic amino acids tend to cluster together in the center of the protein.These will have hydrophobic amino acids on the outside to react with the hydrophobic fatty acids in the membrane.This is not true of integral membrane proteins.These are hydrophilic and will hydrogen bond with water to keep the protein soluble.Amino acids with polar side chains are on outside.
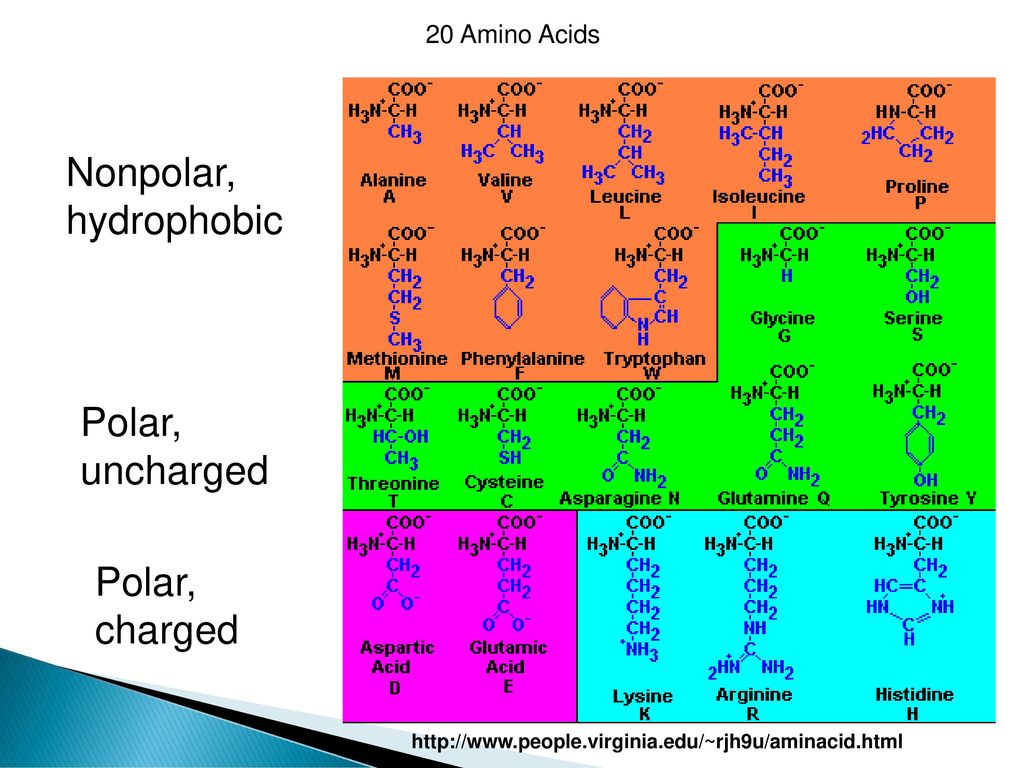
Proteins with either large amounts of cationic or anionic side chains will be longer than proteins carrying a mix of ionic side chains.Some amino acids have bulky side chains that can take up a lot of space.General rules governing interaction of side chains.These interactions include ionic, hydrogen bonds, hydrophobic interactions, and disulfide bonds.
Where secondary structure was a result of hydrogen bonds between peptide groups, tertiary structure is a result of side chains interactions.Tertiary structure involves the three-dimensional folding of a protein due to interactions of amino acid side chains.The amino acid side chains alternate facing above and below the backbone.It is referred to as antiparallel when the regions are going in opposite directions.It is referred to as parallel if both region are going the same direction.Hydrogen bonds form between peptide groups of segments lying adjacent and parallel with one another.The peptide chain is almost completely extended but is folded back on itself.Some amino acids stabilize a -helix while others prevent its formation.The side chains project outwards from the helix.This is tighter than the DNA double-helix.The H of the amine group will hydrogen bond to the oxygen of the carbonyl group three amino acids behind it.The oxygen of the carbonyl group will hydrogen bond to the amine hydrogen three amino acids in front of it.Each peptide group forms 2 hydrogen bonds.The two common secondary structures are the a -helix and the b -sheet.These hydrogen bonds involve only the peptide backbone, not the side chains.Secondary structures are 2-dimensional structures formed due to hydrogen binding between hydrogen of amine groups and oxygen of the carbonyl groups.Amino acid sequence is conventionally written from N-terminus to C-terminus.Proteins have an amino terminal (N-terminus) end and a carboxyl terminal (C-terminus) end.Like DNA with its 5' and 3' end, proteins also have polarity.Peptide bond has partial double bond characteristics therefore it is rigid.Proteins are polymers of amino acids in which the a carbon atoms and peptides alternate forming the backbone and with specific amino acid side chains projecting from the a carbon.Proteins have no biological activity in an unfolded state.A protein is about 3.61 Å long per amino acid in an unfolded state.The primary structure of protein (order of amino acids) determines how the protein folds and interacts at the other levels of interaction.It is generally simpler to sequence DNA than it is to sequence proteins.
#Are hydrophobic amino acids polar or nonpolar code
The amino acid sequence is often deduced from the sequence of nucleotides that code for it.Example: Phe-Ala-Met-Leu-Gln-Trp-Glu-Ile.The primary level of organization is simply the order of amino acids in the peptide chain.Proteins have four level of organization: primary, secondary, tertiary, and quaternary.Proteases or proteolytic enzymes will enzymatically hydrolyze peptides.
#Are hydrophobic amino acids polar or nonpolar free

